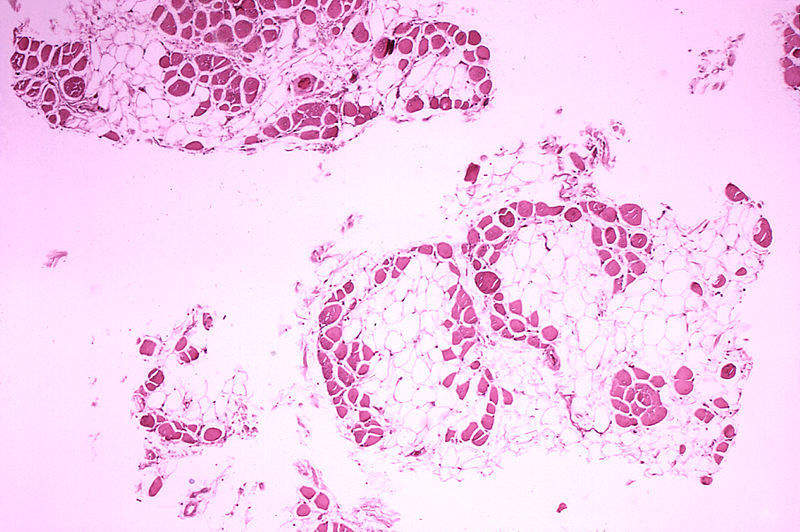
Sarepta Therapeutics is planning to submit a new drug application (NDA) to the US Food and Drug Administration (FDA) for accelerated approval of its therapeutic candidate golodirsen (srp-4053) to treat patients with duchenne muscular dystrophy (DMD).
The move comes after the biopharmaceutical company received guidance on the development pathway for golodirsen from the FDA’s Division of Neurology Products.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Golodirsen is a phosphordiamidate morpholino oligimer being developed for patients with genetic mutations subject to skipping exon 53 of the DMD gene.
Sarepta Therapeutics president and CEO Doug Ingram said: “Obviously, whether golodirsen will obtain accelerated approval is a review decision that will come after the submission and review of our NDA. But we greatly appreciate the willingness of the neurology division to engage and provide clear direction to us on the steps necessary to support an NDA submission for accelerated approval.”
In a Phase I/II clinical trial, statistically significant data was obtained for all biological endpoints following golodirsen treatment in patients with deletions of the DMD gene amenable to exon 53 skipping.
The study was conducted to assess the safety, tolerability, pharmacokinetics and efficacy of golodirsen.
Currently, the efficacy of the drug candidate and casimersen is being investigated in a randomised, double-blind, placebo-controlled Phase III trial (ESSENCE). If the FDA grants accelerated approval, ESSENCE is set to be a post-marketing confirmatory study.
The FDA’s neurology division said that the NDA should include results from long-term animal toxicology studies, which are scheduled to be concluded in the fourth quarter of 2018.




