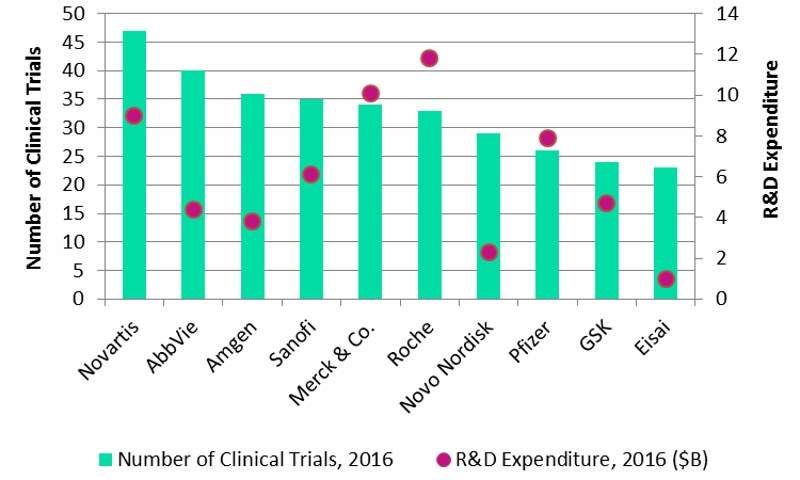
GlobalData’s Clinical Trials Database has identified the top ten pharmaceutical companies in relation to the largest number of active clinical trials in Phase I, II, and III that began during 2016. This is illustrated in the figure below.
However, this is not proportional to the companies’ respective research and development (R&D) expenditure for 2016.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
Figure 1: Number of Active Clinical Trials Versus R&D Expenditure, 2016

Source: GlobalData, Clinical Trials Database
Novartis began the most clinical trials during 2016, which is possibly attributable to the company’s broad pipeline. Unlike its peers, the pipeline covers all therapy areas.
Most pharmaceutical companies studied have restructured their pipelines in recent years to focus their assets on just a handful of core therapy areas. GlaxoSmithKline recently announced it will overhaul its R&D by terminating or selling 13 programmes in clinical development and a larger amount in preclinical development. It will also review options for its rare diseases unit.
Roche and Merck & Co. are the highest R&D spenders in the industry, and have good therapy area coverage with each of their clinical pipelines. This can be attributed to the fact that these companies had the highest proportion of oncology trials.
A total of 64% of Roche’s trials and 56% of Merck’s trials were targeting oncology indications. Roche generated more than half its 2016 sales from this indication and invests considerably in the market. Similarly, Merck is driving a large immuno-oncology development programme and investigating the potential of Keytruda (pembrolizumab) in a range of cancers.
The vast majority of pharmaceutical companies are investing an average of 15% of their top-line revenues on R&D, and some have increased their spending in recent years. However, 22 drugs were approved by the US Food and Drug Administration (FDA) in 2016, which is the lowest number of new drug approvals in the last five years.
This imbalance suggests that a change in the R&D model is required so that research-based pharmaceutical companies can boost innovation and the probability of success of their pipeline assets.




