The Field Guide to Pharmaceutical Powder Characterisation
A variety of parameters influence pharmaceutical powders. Understanding how and why is the first step to optimising your pharmaceutical powder development process.

Anton Paar provides you with a complementary range of solutions for monitoring, investigating, and controlling your products within the pharmaceutical industry – and for optimising your processes and profitability while complying with the regulations.
You have successfully submitted your enquiry. Someone from our company will respond ASAP
Anton Paar develops, produces and distributes highly accurate laboratory instruments and process measuring systems, as well as custom-tailored automation and robotic solutions. We specialise in the measurement of density and concentration of carbon dioxide (CO₂) in rheometry applications.
Our highly precise components, innovative measuring principles and well-designed user interfaces are designed to represent our understanding of quality.
Anton Paar products are created in collaboration with our customers so they can be seamlessly integrated into existing working environments. We aim to provide ideal solutions to meet customers’ requirements.
Innovation is the basis for progress and part of Anton Paar’s DNA, rooted in close cooperation with our customers and outstanding work in the field of research and development (R&D).
For decades, we have invested around 20% of our turnover in R&D. Our R&D team develops new measuring principles, sensor generations and technologies to contribute in the long term to expanding our market leadership and achieving a leading position in other fields.
Using state-of-the-art production technologies, Anton Paar produces high-precision measuring instruments designed to meet customers’ requirements.
The verification and purity control of pharmaceuticals according to their physical properties is essential to ensuring safe medication. Anton Paar’s high-precision instruments are used throughout the pharmaceutical industry for quality control and R&D to ensure production meets the highest standards.
In the highly regulated pharmaceutical sector, quality and safety are the guiding principles. Innovation, high efficiency and developing the highest-quality products are our priorities.
We comply fully with the requirements of pharmacopoeial monographs, current good manufacturing practice (cGMP), US Food and Drug Administration (FDA) 21 Code of Federal Regulations (CFR) Part 11, and EU Annexe 11.
When producing pharmaceutical products, a wide range of parameters need to be monitored, checked and counterchecked.
Anton Paar has numerous solutions for use in quality, production control and R&D. We can also qualify newly purchased instruments on-site and bring them into your workflow within a short period of time.
The resulting test data from clinical trials and their associated metadata are your most valuable assets. These must be collected, processed and stored in accordance with the FDA’s attributable, legible, contemporaneous, original and accurate (ALCOA+) data integrity principles.
The Anton Paar Connect Pharma solution guarantees data integrity through its advanced user management functions, electronic signature settings, and secure date and time-stamped audit trail.
Anton Paar offers several equipment solutions for the pharmaceutical industry. You can assess incoming goods and control final product quality with our density meters, flashpoint testers, refractometers and polarimeters.
Our particle analysers allow you to characterise nanoparticles and microparticles in dispersions and solutions using light-scattering techniques, while our rheometers, viscometers, penetration testers and Zeta potential analysers can investigate the flow behaviour and properties of your product.
Anton Paar’s microwave synthesis solutions enable true scale-up from initial research to full production, and our microwave sample preparation systems are designed for atomic absorption (AA) and inductively coupled plasma (ICP) analysis.
Anton Paar is based in Graz, Austria, and employs more than 4,400 people. We operate six other producing subsidiaries and 40 sales subsidiaries worldwide.
The company’s core competence, high-precision production, combined with its close contact with the scientific community, forms the basis for the quality of its instruments.
Anton Paar is owned by the charitable Santner Foundation.

A variety of parameters influence pharmaceutical powders. Understanding how and why is the first step to optimising your pharmaceutical powder development process.

More than 50 years ago amino acids were found to be an important component in metabolism. That’s why they play a major role in the pharmaceutical and food industry.

Scientists, researchers and industry professionals are invited to attend a free, in-person workshop dedicated to understanding gas physisorption and its vital role in measuring surface area and porosity across a wide range of materials.

Laboratories can now evaluate the Lyza 7000 FTIR spectrometer in their own environment through a new 70-day, no-cost, no-risk trial programme. The initiative allows teams to run real samples, follow existing SOPs, and fully explore the instrument’s capabilities before making a purchase decision.

Anton Paar introduces the PNR 500, an advanced automatic penetrometer designed for fast, reproducible consistency and penetration testing of semi-solid materials.

Anton Paar has announced the launch of the Litesizer DLS 301, the latest addition to its established Litesizer family.

Anton Paar offers a new era of quality control rheometry with the launch of the MCR 53, MCR 73, and MCR 93.

Anton Paar has launched its new line of Xsample sample changers for handling low-viscosity samples.

Anton Paar Ltd, a global leader in high-precision laboratory and process instrumentation, has launched a limited-time trade-in programme offering customers in the UK up to 30% off the price of new instruments across its comprehensive product portfolio.

Anton Paar has launched the Julia DSC series, a powerful new generation of differential scanning calorimeters (DSC) developed to deliver unmatched speed and simplicity for materials analysis.

Anton Paar presents a significant addition to the renowned and proven Ultrapyc series of gas pycnometers. The two new additions, the Ultrapyc 7000 and Ultrapyc 7000 Micro - deliver high throughput with advanced automation and precision for small samples.

Building on its industry-leading legacy in rheometry, Anton Paar has unveiled its next generation of modular compact rheometers (MCRs), setting new benchmarks for precision, speed, intelligence, and adaptability in material characterisation.

Anton Paar announces the launch of its new Thermal Analyser, hosted under the theme 'Where Speed Meets Precision.'

This new generation of instruments enables users to achieve greater precision, streamline workflows, and unlock a deeper understanding of material behaviour.

Anton Paar, a world market leader in density measurement, has launched a new generation of digital density meters.

The highly anticipated Basics of Rheology Workshop will take place on July 30th and 31st, 2025, at Anton Paar Ltd. in Luton.

Anton Paar proudly introduces the L-Rix 2100 and L-Rix 3100, innovative inline refractometers designed for continuous Brix concentration monitoring in metalworking machines, industrial processes, and fruit and vegetable processing. With their unique combination of high precision, compact design, and affordability, these instruments set a new standard in reliable process control for industrial applications.

Anton Paar’s Abbemat Pharma refractometers are the ideal solution for laboratories in the pharmaceutical industry, delivering precise refractive index (RI) measurements while ensuring full compliance with regulatory requirements, eg. US pharmacopeia . Designed to meet the demands of highly regulated environments, these instruments provide reliable performance, robust data management, and audit-readiness from the outset, making them an indispensable tool for pharmaceutical quality control.

This workshop is aimed at those who are looking to find a solution to their viscosity measurement or customers who are new to viscosity. The workshop will involve theory and practical sessions.

Anton Paar introduces SAXSpoint 500 and SAXSpoint 700, state-of-the-art laboratory beamlines offering unparalleled capabilities in SAXS/WAXS/GISAXS/USAXS/RheoSAXS analysis. Designed to meet the needs of researchers and industrial users, SAXSpoint instruments combine precision, flexibility, and efficiency, enabling comprehensive material studies under diverse experimental conditions.

Join us for a comprehensive FREE two-day seminar and workshop at our Luton office (can opt to join day 1 and/or 2) dedicated to exploring two pivotal techniques in surface chemistry and material science: physisorption and chemisorption.
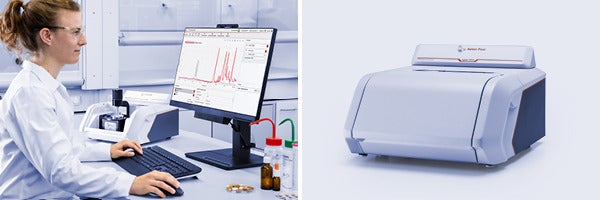
Lyza 3000 is the latest addition to Anton Paar’s industry-transforming Lyza series, offering a budget-friendly yet advanced FTIR spectrometer for comprehensive molecular analysis. This cutting-edge instrument simplifies material identification and verification.

Anton Paar announces the launch of Litesizer DIF 500, a laser diffraction particle size analyser designed to deliver accurate particle size measurements in high-tech laboratories and even challenging industrial environments. With the ability to measure samples ranging from 0.01µm to 3,500µm, Litesizer DIF 500 stands out for its precision and durability.

Anton Paar is proud to announce the new Litesizer DLS, a groundbreaking dynamic light scattering (DLS) instrument that sets new standards for particle analysis. Designed for precision, reliability and user convenience, the Litesizer DLS combines advanced features with intuitive operation, making it the ideal choice for laboratories seeking high-quality analytical results. Measure particles in the size range of 0.3nm to 12µm.

Perfect sample preparation leads to superior trace elemental analysis.

Anton Paar is proud to introduce the Ultratap 500 series, an innovative leap in tap density testing. Engineered for durability, these instruments guarantee 25 million taps without calibration loss, setting a new industry standard. Backed by a robust three-year warranty, the Ultratap 500 series assures reliable performance for years, making it the most dependable choice for tap density analysis.

For gas adsorption experiments, the quality of nitrogen gas used is key for getting repeatable results. Unfortunately, nitrogen is notoriously difficult to ship and set up in a lab.

Anton Paar, a leading expert in precision instruments for material characterisation, has announced the launch of a new accessory for existing rheometers, heralding a cost-effective solution for analysing envelope density. This latest innovation promises to enhance production, as well as characterisation and quality control of solids throughout various industries by offering automated, standards-compliant analysis capabilities.

Introducing the latest version of AP Connect, a laboratory software that connects instruments from Anton Paar and other vendors, communicates measurement information, automates task execution and ensures compliance. Making research easier than ever before, the lab execution software is designed for straightforward operation with its plug and play capabilities.
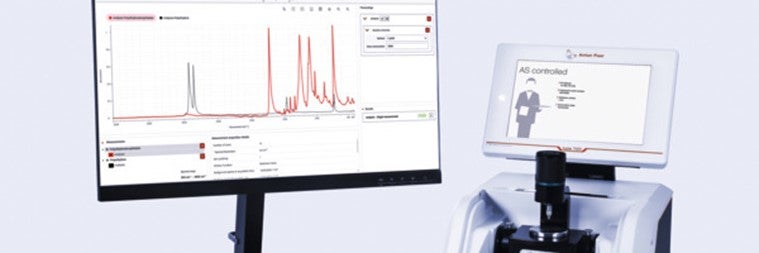
Anton Paar, a leader in precise laboratory instruments, announces the launch of the Anton Paar Spectroscopy Suite for Lyza 7000 FTIR spectrometers.

Anton Paar’s exceptional Step series brings a new standard when it comes to mechanical surface testing platforms, guaranteeing higher precision and savings in time and resources.

Do you need to measure wet and dry dispersions with high throughput? Then check out our new Autosampler.
Anton Paar develops, produces and distributes highly accurate laboratory instruments and process measuring systems, and provides custom-tailored automation and robotic solutions. It is the world leader in the measurement of density, concentration and CO₂ and in the field of rheometry, and is well-placed to serve the needs of analytical scientists in the modern pharmaceutical industry.